2016 Overview of HTA reports for Medical Devices and In-Vitro Diagnostics
09 Feb 2017
HTA plays an increasingly important role for Medical Device and IVD companies. Below is a summary of the HTA Dynamics during 2016, followed by the same data for 2015.
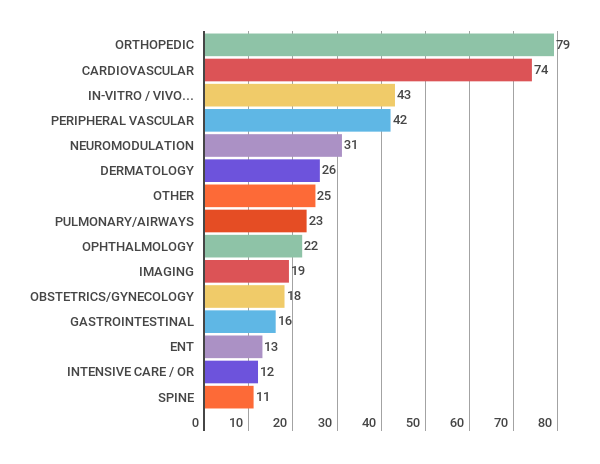
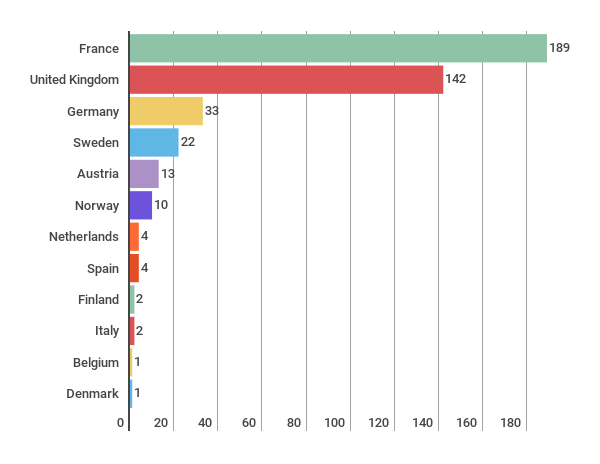
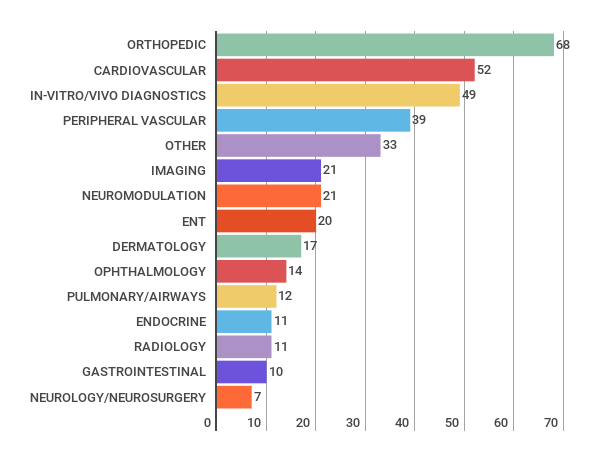
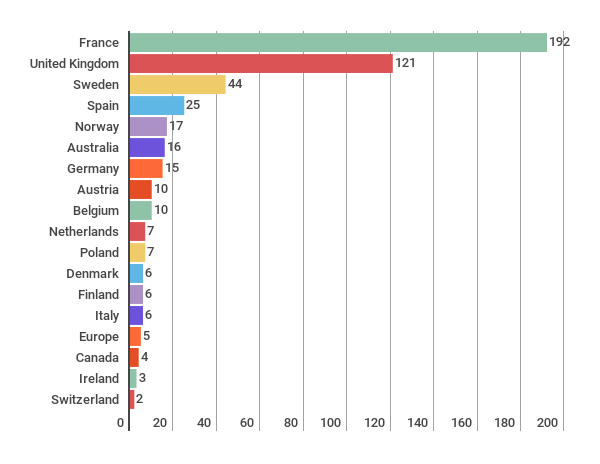
The total amount of HTA evaluations have reduced somewhat from 490 (2015) to 427 (2016). Orthopedics continues to be the most common topic of the evaluations.
2016 overview
Number of reports per country
Number of reports per technology area
2015 overview
Number of reports per country
Number of reports per technology area